|
This article was authored by Will Daughton and originally published on Pickleball.com here. DALLAS, TX - Though there aren’t any more professional pickleball events in 2023, a few pros are still finding ways to end the year with some friendly competition. Zane Navratil and Altaf Merchant are leading a fundraiser in collaboration with Austin-based nonprofit Stop Alzheimer’s Now (SAN) to see who can raise the most money for Alzheimer’s research. The contest also features experiential prizes for donors that go above and beyond, including free admission to a clinic led by Zane and Altaf in 2024, and the opportunity to partake in a pro-am match with both of them. So far, the pair have raised over $37,000 since the month-long competition began Nov. 28. Teaming up with a nonprofit for Alzheimer’s research has been a goal of Navratil’s since witnessing his grandmother battle the disease. “Pretty much since I went into pro pickleball in 2020, I wanted to find one that I could give back to and use my fan base to support,” he shared. He wanted to find an organization where his entire donation would go towards the cause and not be split between research efforts and organizational expenses. When he met SAN founder Shaun McDuffee in early 2023, the conversation quickly turned to the nonprofit. Zane was interested in learning more, but he wanted to inspect the organization’s books before making a commitment. The former auditor was impressed with what he found. “There are zero expenses other than the ones required by law, which is great because when I give a dollar, I want to know that a dollar is actually going to the cause,” he said. Navratil has been a SAN ambassador ever since and paved the way for other pickleball players like Merchant, Jayson Harris, and Hartland Jones to join the effort. Having seen the ruthlessness of the disease affect his own father, Merchant reached out to McDuffee about getting involved in September. Zane and Altaf’s fundraiser is just the latest step that SAN has taken with the pickleball community. McDuffee discovered pickleball in 2021, eight years after he founded SAN alongside his wife Kristin. For much of those first eight years, he focused on projects like walking over 1,000 miles across 17 states to raise awareness about the disease. Naturally, he fell in love with the sport, and he saw a potential vessel through which SAN’s impact could grow. “Rotary clubs are credited with helping cure polio because it was a bunch of business owners who got together and then these clubs started raising money for it,” he explained. “I thought, ‘Man, how cool would it be if pickleball could be associated with curing Alzheimer’s?’"
McDuffee has been working to bring that idea closer to reality over the past year. “I started connecting the dots in 2022 and realized, ‘Man, this is a perfect fit,’” he said. He began networking with players and figures in the picklesphere while attending different events and tournaments in 2023, and he’s grateful that he keeps finding individuals across the sport who continue to be invested in Stop Alzheimer’s Now. “I just think it shows that this sport is more than just a sport,” he said. “It’s a community of people that care.” To fully immerse himself in the giving spirit of the holidays, McDuffee will also match the total amount of money that is donated to Zane and Altaf. The Year-End Big Give runs through Dec. 28.
0 Comments
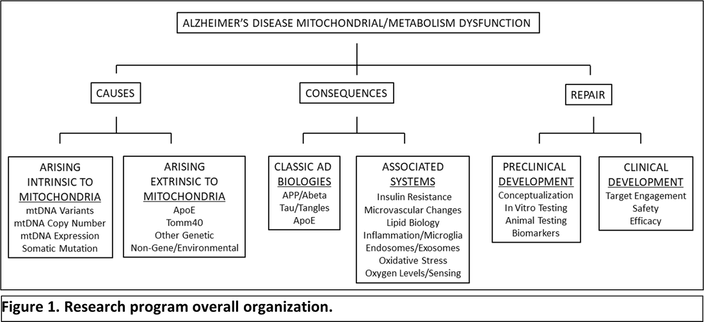
2023 Update: SAN's contributions to to the University of Kansas Alzheimer's Disease Research Center10/6/2023 In 2016, Stop Alzheimer’s Now contributed $100,000 to the University of Kansas Alzheimer’s Disease Center, establishing the Stop Alzheimer’s Now Research Fund. The donation was made to support a ketogenic diet study conducted by Dr. Russell Swerdlow and his team, including Scott Koppel, M.D., Ph.D., as they studied how ketone body-based interventions affect the prevention and treatment of Alzheimer’s disease and the decisions of physicians and patients regarding treatment options. We recently received an update from Dr. Swerdlow on how SAN's donation was used toward research, as well as recent work from his lab as they continue to search for an Alzheimer's cure. Your support played an outsize role in some of my recent successes. It really allowed me to play jazz with my research, and to take some chances. In particular, SAN dollars were used to support work done by a trainee, graduate student now turned MD PhD, named Scott Koppel. Stop Alzheimer’s Now support allowed us to generate a unique transgenic mouse, which unfortunately did not turn out to have a detectable phenotype. While that was disappointing to us, this is not an uncommon outcome of such experiments and we could not have made progress on this, and addressed the question we needed to answer, without the contribution. Your support did help make possible additional thesis work by Dr. Koppel, and the Foundation is listed as a key sponsor of a publication I am very proud of, and which I think is a very important paper.” The full publication on this study can be found here: A Ketogenic Diet Differentially Affects Neuron and Astrocyte Transcription. Thank you to Dr. Swerdlow and the KU Alzheimer's Research Disease Center for their dedication and groundbreaking research toward finding a cure for Alzheimer's. For a summary of recent work from Dr. Swerdlow's lab, click here. Russell H. Swerdlow, MD Gene and Marge Sweeney Professor Professor of Neurology, Cell Biology and Physiology, Biochemistry & Molecular Biology Director, University of Kansas Alzheimer's Disease Research Center Director, KUMC Neurodegenerative Disorders Program University of Kansas School of Medicine Dr. Swerdlow continues his research into Alzheimer’s disease (AD) mitochondrial dysfunction. Studies focus on genes that underly mitochondrial function including mitochondrial DNA (mtDNA) and the APOE gene; mitochondrial connections to the classic AD pathologies including amyloid plaques and tau tangles; and how to manipulate mitochondria to treat AD patients. Efforts advance Dr. Swerdlow’s “Mitochondrial Cascade Hypothesis,” which uniquely proposes AD arises as a primary consequence of mitochondrial stress. According to this novel paradigm, mitochondria initiate AD, drive its progression and pathologies including plaques and tangles, and provide a roadmap for developing effective therapies. Figure 1 illustrates the program’s overall organization: Stop Alzheimer’s Now support was especially critical in supporting research performed by one of Dr. Swerdlow’s graduate students, Scott Koppel MD PhD. Dr. Koppel is now pursuing his neurology residency training at Cedars Sinai Medical Center in Los Angeles. Stop Alzheimer’s Now is acknowledged as a major benefactor of the following publication, which found that a ketogenic diet affects the brain in ways that may be expected to counter brain changes that are seen in Alzheimer’s disease patients:
Of note, in June 2022 Dr. Swerdlow received an international award, the Oskar Fischer Prize. The awarding committee recognized the Mitochondrial Cascade Hypothesis as one of the world’s most promising attempts to conceptualize AD. In the past year Dr. Swerdlow delivered invited Mitochondrial Cascade Hypothesis national and international presentations to these forums:
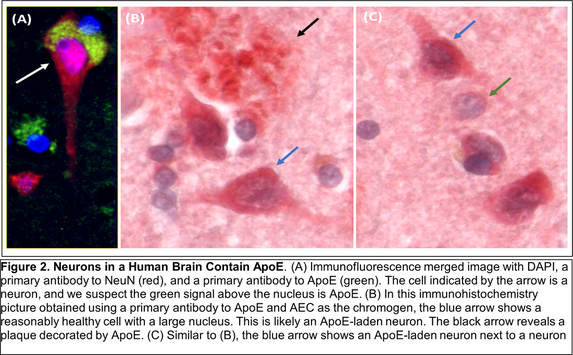
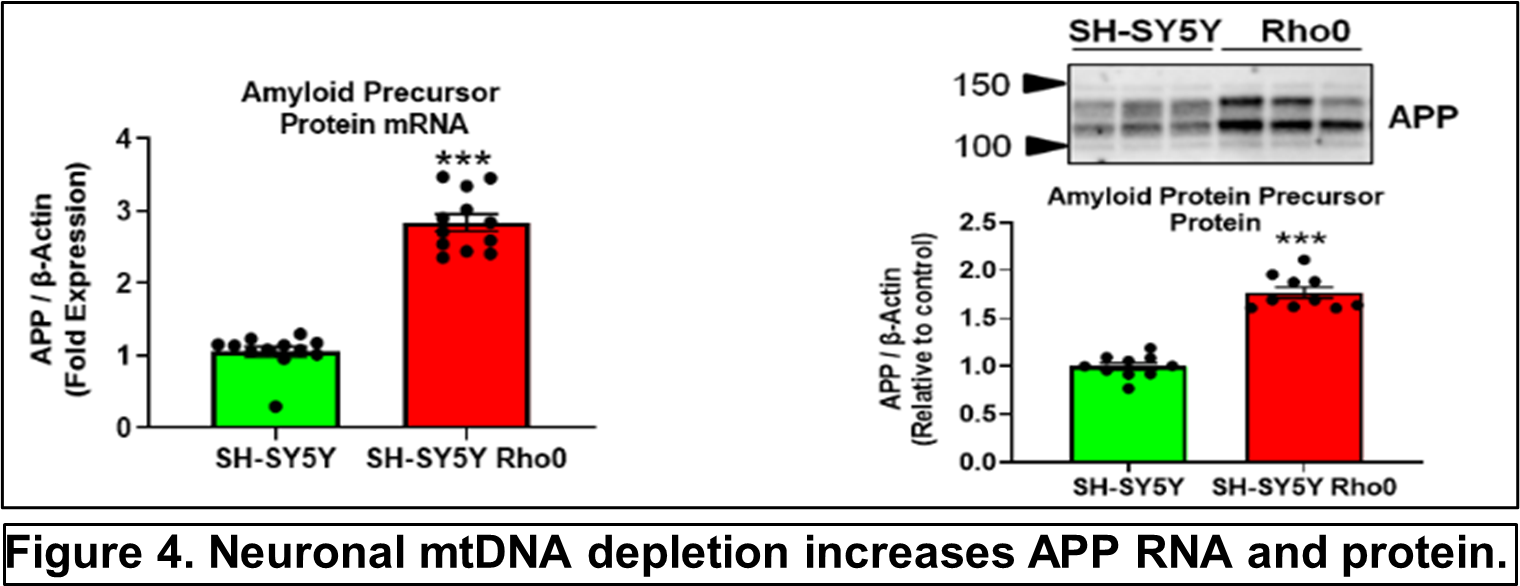
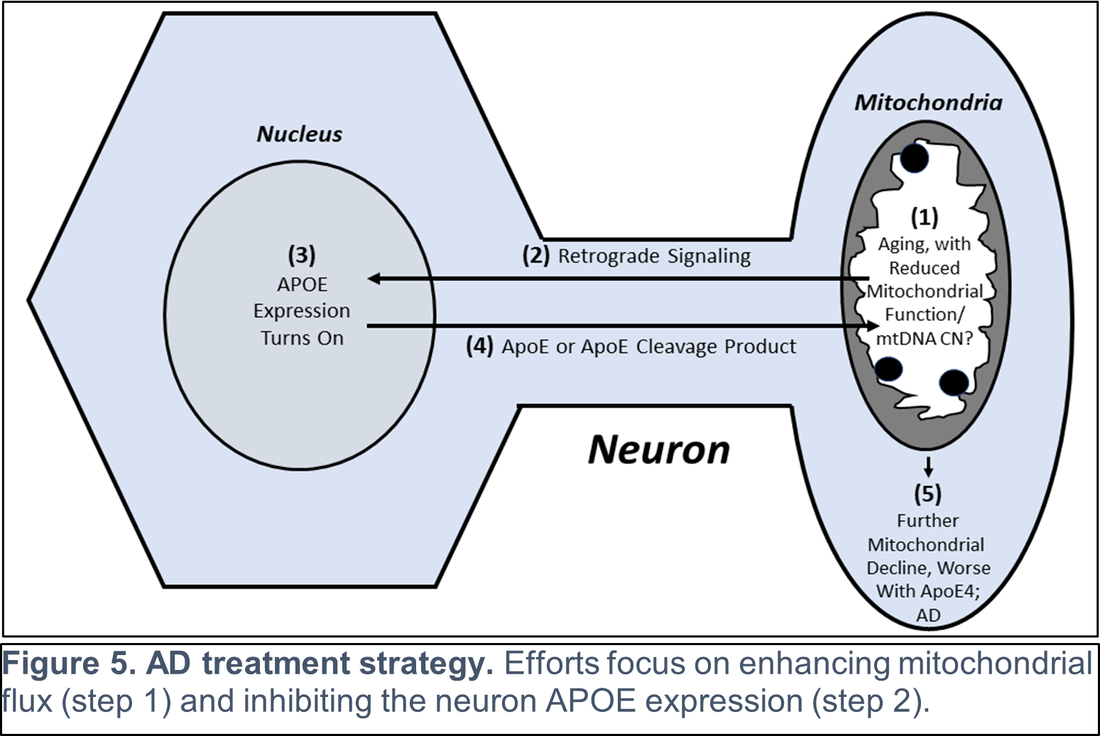
Other recent publications can be found here. Specific recent major accomplishments, and immediate directions, are discussed below. Genetic Studies We reported mitochondrial DNA (mtDNA) copy number was lower in brains of persons who died with AD than it was in the brains of persons who did not. We further demonstrated inverse associations between mtDNA copy number and AD histology changes (plaques and tangles) and the magnitude of cognitive impairment within an individual. Essentially, brains containing less mtDNA contained more plaque and tangle pathology, and persons with less mtDNA had more cognitive impairment. Some investigators will assume reduced mtDNA copy number is a consequence and not a cause of AD. We suspect it is more likely a cause and data we generated support this view. We found a specific heritable mtDNA sequence, haplogroup J, associates with higher mtDNA copy number and increased plaque/tangle pathology. Haplogroup J carriers also show evidence of reduced late-life memory capacity, which manifests prior to AD symptoms. In a related study, we observed aging mice normally upregulate their brain mtDNA copy number. Given this perspective, we propose that to maintain normal function neurons containing haplogroup J mtDNA must maintain a higher baseline mtDNA copy number, and as those neurons age their capacity to mount an age-determined compensatory increase in their mtDNA is consequently diminished. The APOE gene that encodes the apolipoprotein E protein associates with AD for unclear reasons. We find that in settings of mitochondrial dysfunction, neurons turn on APOE gene expression. This is important because neurons normally do not express APOE. We subsequently examined AD autopsy brains to look for evidence of neuron APOE expression and found APOE-laden neurons (Figure 2). This is remarkable because APOE is a secreted protein, so in addition to aberrantly expressing APOE the neurons appear unable to properly secrete it. Linking Mitochondria to AD Pathologies Our recently published data show when neuronal cells express the amyloid precursor protein (APP), only a small amount localizes to mitochondria. Most APP distributes to the plasma membrane. Neurons process APP to beta amyloid (Aβ), which they secrete. We found that in settings of mitochondrial dysfunction, APP redirects to mitochondria, less reaches the plasma membrane, and less Aβ secretion occurs. We believe this can at least partly explain why cerebrospinal (CSF) Aβ levels are reduced in AD patients. We in fact believe CSF levels can serve as a biomarker of brain mitochondrial function. To address this hypothesis, we recently began obtaining CSF from comatose patients hospitalized in the KU Hospital intensive care unit with traumatic brain injuries or subarachnoid hemorrhage. Brain mitochondria in comatose patients are relatively inactive, but activity returns as the coma resolves. We are testing for associations between level of consciousness and Aβ in these samples. We are also measuring the tangle-forming protein tau, as well as mitochondrial components including mtDNA. Through this we hope to develop CSF assays that will inform the status of a living person’s brain mitochondria without having to perform an actual brain biopsy. We are further working to link mitochondrial and APOE biology. We are interested in how neuronal apolipoprotein E impacts overall neuron integrity, and directly or indirectly affects mitochondrial function. We therefore created neuronal cell lines that uniquely express the different APOE2, 3, and 4 isoforms. We predicted APOE4, the isoform that associates with increased AD risk, would disproportionally impair mitochondria but surprisingly discovered constitutive expression of all APOE isoforms induces equivalent states of mitochondrial stress. The apolipoprotein E protein plays a recognized role in lipid biology, which is broadly altered in AD patients. Accordingly, we initiated a “lipidomics” study to determine how mitochondrial dysfunction alters neuronal cell lipidomes. Mitochondrial and Metabolism-Targeted AD Therapeutics We recently enrolled our 30th participant into a randomized trial that is evaluating how a ketogenic diet affects cognitive abilities in AD patients. Our goal is to is matriculate 80 participants. The rationale is that inducing ketone body production through a ketogenic diet will partly compensate for the state of reduced glucose utilization we know exists in AD. We believe this state reflects a consequence of mitochondrial dysfunction, and that adding the ketone fuel will enhance the brain’s ability to generate energy and signals that will promote mitochondrial maintenance. As part of this study, we perform extensive assays using blood cells from our trial participants. This includes applying a powerful omics technique, RNASeq, to determine how a ketogenic diet effects overall gene transcription in the trial participants. This past year we also completed a study assessing the impact of a diabetes drug, dapagliflozin, on AD patients. Dapaglifozin, an SGLT2 inhibitor, blocks the local re-uptake of renally excreted glucose. This leads to a mild reduction in blood glucose levels and a mild state of ketosis. We are currently analyzing the dapagliflozin trial data. In 2020 we reported that administering adequate doses of the metabolism intermediate oxaloacetate (OAA), a dicarboxylic acid, can promote brain bioenergetic fluxes in human AD patients. Because OAA is unstable in solution, we created OAA prodrugs in which OAA’s carboxyl ends are esterified to a ketone body, β-hydroxybutyrate, or a lactate precursor, propylene glycol. The former molecule was designed to deliver OAA as part of a ketogenic diet mimetic intervention, and the latter to deliver OAA as part of an exercise mimetic intervention. We are currently determining oral absorption, tissue distribution, and pharmacokinetics in the preclinical setting. These studies, which required us to generate isotope-labeled versions of the compounds, are intended to move us towards investigational new drug application (IND) filings with the FDA. Support from Stop Alzheimers Now dramatically accelerated the pace and elevated the quality of Dr. Swerdlow’s research program. Since his initial support, the lab has enhanced its international recognition and reputation. Stop Alzheimers Now support was instrumental to our successes and is sincerely appreciated.
POSTED ON JANUARY 07, 2020 The Lewis Katz School of Medicine at Temple University (LKSOM) has received a gift from Temple University trustee, benefactor and Fox School of Business alumnus Phil Richards and the Scott Richards North Star Charitable Foundation (SRNSCF), as well as Stop Alzheimer’s Now (SAN), to establish the Scott Richards North Star Foundation and Stop Alzheimer’s Now Chair for Alzheimer’s Research at the Alzheimer’s Center at Temple. The gift to establish the Chair will be utilized exclusively for the purpose of funding Alzheimer’s research at the Alzheimer’s Center at Temple (ACT). The name of the researcher who will be first occupant of the new chair position will be announced at a later date. “We’re extremely thankful to the Scott Richards North Star Charitable Foundation and Stop Alzheimer’s Now for this generous gift,” said John M. Daly, MD, FACS, Interim Dean and Harry C. Donahoo Professor of Surgery at LKSOM. “That these two organizations would select Temple for this donation speaks volumes to the promising work being done by Dr. Domenico Praticò and his team at the Alzheimer’s Center at Temple. The Alzheimer’s Center is performing cutting-edge research into the causation, prevention and ultimately cure of this devastating disease.” The Scott Richards North Star Charitable Foundation is a Minnesota non-profit corporation, and Stop Alzheimer’s Now is a Texas non-profit corporation. Both have a core funding priority of ending Alzheimer’s disease. In 2018 SRNSCF provided a gift to LKSOM to establish the Scott Richards North Star Charitable Foundation Chair for Alzheimer’s Research at Temple as well as the Alzheimer’s Center at Temple. Domenico Praticò, MD, was appointed the first Scott Richards North Star Charitable Foundation Chair for Alzheimer’s Research and Professor and Director of the Alzheimer’s Center at Temple. The occupant of the new chair funded by this latest gift will report to Dr. Praticò. SAN also provided a gift in early 2019 to fund research at ACT. The Next Leg of our Journey |